|
9/4/2023 0 Comments Negative electron affinity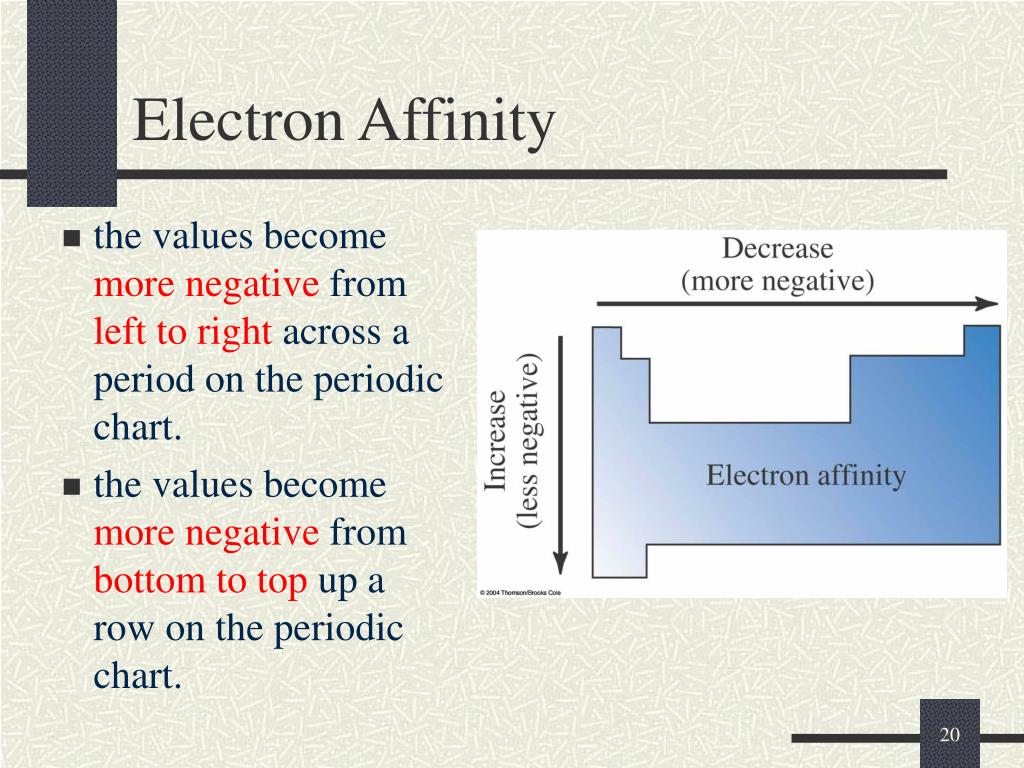
The chemical rationale for changes in electron affinity across the periodic table is the increased effective nuclear charge across a period and up a group.Įlectron affinity: The electron affinity of an atom or molecule is defined as the amount of energy released when an electron is added to a neutral atom or molecule to form a negative ion.Įlectronegativity: The tendency of an atom or molecule to attract electrons to itself.Electron affinity generally increases across a period in the periodic table and sometimes decreases down a group. There are general trends in electron affinity across and down the periodic table of elements.The electron affinity of an atom or molecule is the propensity for that particle to gain an electron.Electron Affinity TrendsĪpplications of Hard-Soft Acid-Base theoryĬhemistry Question Pack Passage 1 Question 4 Electron affinity follows the trend of electronegativity: fluorine (F) has a higher electron affinity than oxygen (O), and so on. However, this trend applies only to Group-1 atoms. Since this electron is farther away, it should be less attracted to the nucleus and release less energy when added. A trend of decreasing electron affinity down the groups in the periodic table would be expected since the additional electron is entering an orbital farther away from the nucleus. Chlorine has the highest electron affinity while mercury has the lowest.Įlectron affinity generally increases across a period (row) in the periodic table, due to the filling of the valence shell of the atom. The electron affinities of the noble gases have not been conclusively measured, so they may or may not have slightly negative values. Atoms, such as Group 7 elements, whose anions are more stable than neutral atoms have a higher electron affinity. Please notify me of errors/problems Negative charges on attached groups. 
Generally, nonmetals have a more positive electron affinity than metals. The electron affinity is a) the energy required to remove an electron from an. This can be shown for the chloride ion formation below: The electron affinity (E ea) of a neutral atom or molecule is defined as the amount of energy released when an electron is added to it to form a negative ion. Electron affinity is the energy change that occurs when a neutral atom gains an electron.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
